CHEMICAL BONDING
by Mark Lloyd Coloma
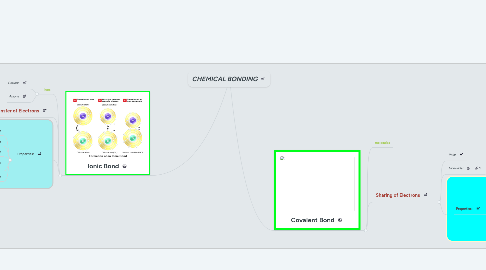

1. Ionic Bond
1.1. ions
1.1.1. Cations
1.1.2. Anions
1.2. Transfer of Electrons
1.3. Properties:
1.3.1. formed between metals and non-metals
1.3.2. Dissolve easily in water
1.3.3. Easily conducts electricity in a solution
1.3.4. Form crystalline solids in high melting temperatures
1.3.5. Have higher melting and boiling points
2. Covalent Bond
2.1. molecules
2.2. Sharing of Electrons
2.2.1. Polar
2.2.2. Non-Polar
2.2.3. Properties:
2.2.3.1. Formed between nonmetals
2.2.3.2. Not soluble in water
2.2.3.3. Don't conduct electricity in water
2.2.3.4. Are soft and squishy
2.2.3.5. Have lower melting and boiling points
