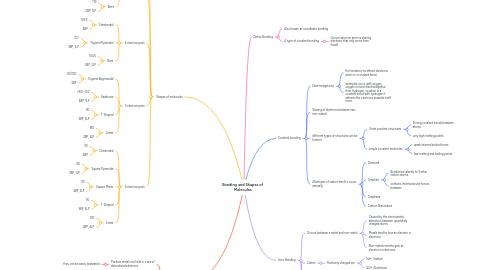
1. Covalent bonding
1.1. Electronegativity
1.1.1. the tendency to attract electrons when in a covalent bond.
1.1.2. examples occur with oxygen. oxygen is more electronegative than hydrogen, so when in a covalent bond with hydrogen it attracts the electrons towards itself more.
1.2. Sharing of electrons between two non-metals
1.3. different types of structures can be formed
1.3.1. Ginat covalent structures
1.3.1.1. Strong covalent bonds between atoms
1.3.1.2. very high melting points
1.3.2. simple covalent molecules
1.3.2.1. weak intermolecular forces
1.3.2.2. low melting and boiling points
1.4. Allotropes of carbon tend to occur naturally
1.4.1. Diamond
1.4.2. Graphite
1.4.2.1. Bonded covalently to 3 other carbon atoms.
1.4.2.2. contains intermolecular forces between
1.4.3. Graphene
1.4.4. Carbon Nanotubes
2. Ionic Bonding
2.1. Occurs between a metal and non-metal
2.1.1. Caused by the electrostatic attraction between oppositely charged atoms
2.1.2. Metals tend to lose an electron or electrons
2.1.3. Non-metals tend to gain an electron or electrons
2.2. Cation
2.2.1. Positively charged ion
2.2.1.1. Na+, Sodium
2.2.1.2. Al3+, Aluminium
2.3. Anion
2.3.1. Negatively charged ion
2.3.1.1. O2-, Oxygen
2.3.1.2. Cl-, Chlorine
2.4. Many compounds can also have charges
2.4.1. NH4+, Ammonium Ion
2.4.2. SO4 2-, Sulphate Ion
3. Dative Bonding
3.1. Also known as coordinate bonding
3.2. A type of covalent bonding
3.2.1. Occurs when an atom is sharing electrons that only come from itsself
4. Shapes of molecules
4.1. 2 electron pairs
4.1.1. Linear
4.1.1.1. 180
4.1.1.2. 2BP
4.2. 3 electron pairs
4.2.1. Trigonal Planar
4.2.1.1. 120
4.2.1.2. 3BP
4.2.2. Bent
4.2.2.1. 119
4.2.2.2. 2BP, 1LP
4.3. 4 electron pairs
4.3.1. Tetrahedral
4.3.1.1. 109.5
4.3.1.2. 4BP
4.3.2. Trigonal Pyramidal
4.3.2.1. 107
4.3.2.2. 3BP, 1LP
4.3.3. Bent
4.3.3.1. 104.5
4.3.3.2. 2BP, 2LP
4.4. 5 electron pairs
4.4.1. Trigonal Bipyramidal
4.4.1.1. 90/120
4.4.1.2. 5BP
4.4.2. Sawhorse
4.4.2.1. <90/<120
4.4.2.2. 4BP, 1LP
4.4.3. T-Shaped
4.4.3.1. 90
4.4.3.2. 3BP, 2LP
4.4.4. LInear
4.4.4.1. 180
4.4.4.2. 2BP, 3LP
4.5. 6 electron pairs
4.5.1. Octahedral
4.5.1.1. 90
4.5.1.2. 6BP
4.5.2. Square Pyramidal
4.5.2.1. ~90
4.5.2.2. 5BP, 1LP
4.5.3. Square Planar
4.5.3.1. 90
4.5.3.2. 4BP, 2LP
4.5.4. T-Shaped
4.5.4.1. 90
4.5.4.2. 3BP, 3LP
4.5.5. Linear
4.5.5.1. 190
4.5.5.2. 2BP, 4LP
5. Metallic Bonding
5.1. Positive metal ions held in a sea of delocalised electrons
5.1.1. they can be easily separated
5.2. Properties
5.2.1. HIgh melting and boiling points
5.2.1.1. strong metallic bonds present
5.2.2. conductors of heat and electricity
5.2.2.1. delocalised electron can carry the charge
5.2.3. Malleable
5.2.3.1. Atoms are arranged in such a way that they can slide over each other
