All chemical reazctions
von Mia Harden
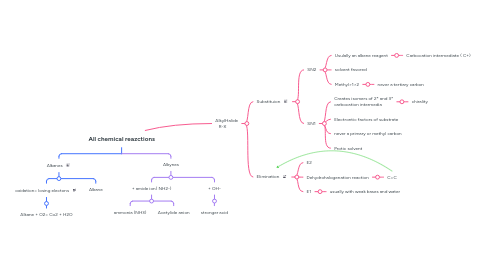

1. Alkanes
1.1. oxidation= losing electons
1.1.1. Alkane + O2= Co2 + H2O
1.2. Alkane
2. Alkynes
2.1. + amide ion( NH2-)
2.1.1. ammonia (NH3)
2.1.2. Acetylide anion
2.2. + OH-
2.2.1. stronger acid
3. AlkylHalide R-X
3.1. Substituion
3.1.1. SN2
3.1.1.1. Usulally an alkene reagent
3.1.1.1.1. Carbocation intermediate ( C+)
3.1.1.2. solvent favored
3.1.1.3. Methyl>1>2
3.1.1.3.1. never a tertiary carbon
3.1.2. SN1
3.1.2.1. Creates isomers of 2* and 3* carbocation intermedia
3.1.2.1.1. chirality
3.1.2.2. Electrontic factors of substrate
3.1.2.3. never a primary or methyl carbon
3.1.2.4. Protic solvent
3.2. Elimination
3.2.1. E2
3.2.2. Dehydrohalogenation reaction
3.2.2.1. C=C
3.2.3. E1
3.2.3.1. usually with weak bases and water
