1. Atom: The smallest unit of matter or an element.
1.1. Subatomic Particles: A particle that is smaller than an atom.
1.1.1. Protons: Particle with positive electric charge.
1.1.2. Neutrons: Particle without an electric charge.
1.1.3. Electrons: Particle with negative electric charge.
2. Types of matter
2.1. Solids: Firm and stable, isn’t fluid.
2.1.1. Metals: Are solids, conductors, and malleable.
2.1.2. Metalloids: Have characteristics of both metals and non-metals.
2.2. Gases: Expands, doesn’t have a fixed shape or volume.
2.2.1. Non-metals: are typically gases.
2.3. Liquids: Has a constant volume but flows freely.
3. Types of matter 2
3.1. Pure Substances: are made of only one kind of particle and has a constant composition.
3.1.1. Ex. water, sugar, salt, etc.
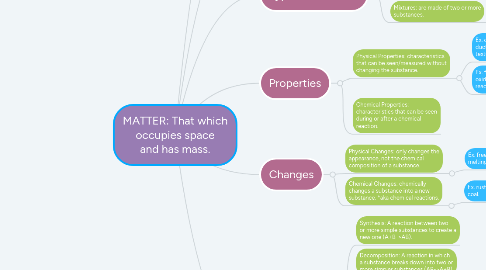
3.2. Mixtures: are made of two or more substances.
3.2.1. Ex. salt water, orange juice, brewed tea, etc.
4. Properties
4.1. Physical Properties: characteristics that can be seen/measured without changing the substance.
4.1.1. Ex. colour, density, volume, mass, ductility, boiling/melting, malleability, texture, etc.
4.1.2. Ex. flammability, toxicity, solubility, oxidation, corrosion, water/acid reactivity, etc.
4.2. Chemical Properties: characteristics that can be seen during or after a chemical reaction.
5. Changes
5.1. Physical Changes: only changes the appearance, not the chemical composition of a substance.
5.1.1. Ex. freezing/boiling water and melting/cooling wax.
5.2. Chemical Changes: chemically changes a substance into a new substance. *aka chemical reactions.
5.2.1. Ex. rusting of metal and burning of coal.
6. Chemical Reactions: change of the molecular/ionic structure of a substance.
6.1. Synthesis: A reaction between two or more simple substances to create a new one (A+B->AB).
6.2. Decomposition: A reaction in which a substance breaks down into two or more simpler substances (AB->A+B).
6.3. Single-displacement: A reaction in which one element of a compound is replaced by another (A+BX->B+AX).
6.4. Double-displacement: A reaction in which two compounds exchange ions to create two new compounds (AY+BX->BY+AX).
6.5. Combustion: A reaction in which a substance reacts with oxygen and release energy in the form of heat/light/fire.
6.6. Acid-Base: A reaction in which a hydrogen ion is donated by the acid and accepted by the base.
6.6.1. Acid: A substance that donates protons and accepts a pair of valence electrons to form a bond.
6.6.2. Base: A substance that donates a pair of valence electrons to form a bond and accepts protons.
6.6.3. pH: A scale that measures how acidic/basic a liquid substance is. The scale is between 0-14. <7 is acidic and >7 is basic.
