Microbiologia Industrial
Door Gabriela Colon
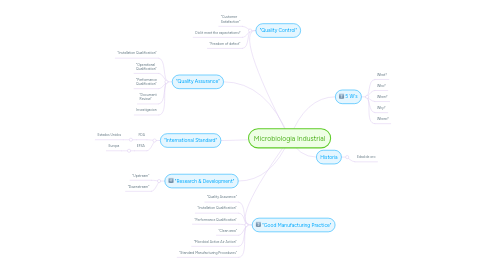

1. "Good Manufacturing Practice"
1.1. "Quality Assurance"
1.2. "Installation Qualification"
1.3. "Performance Qualification"
1.4. "Clean area"
1.5. "Microbial Active Air Action"
1.6. "Standard Manufacturing Procedures"
2. "Research & Development"
2.1. "Upstream"
2.2. "Downstream"
3. "International Standard"
3.1. FDA
3.1.1. Estados Unidos
3.2. EFSA
3.2.1. Europa
