chemical bonding
by lam man ching
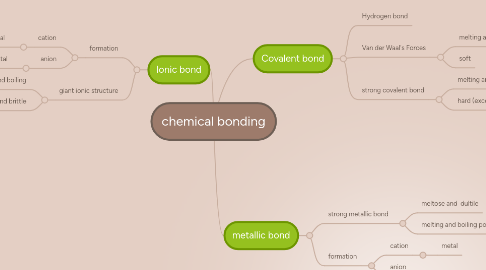

1. Ionic bond
1.1. formation
1.1.1. cation
1.1.1.1. non-metal
1.1.2. anion
1.1.2.1. metal
1.2. giant ionic structure
1.2.1. melting and boiling
1.2.1.1. high
1.2.2. hard and brittle
2. Covalent bond
2.1. Hydrogen bond
2.2. Van der Waal's Forces
2.2.1. melting and boiling point
2.2.1.1. low
2.2.2. soft
2.3. strong covalent bond
2.3.1. melting and boiling point
2.3.1.1. very high
2.3.2. hard (except graphite)
3. metallic bond
3.1. strong metallic bond
3.1.1. meltose and dultile
3.1.2. melting and boiling point
3.1.2.1. very high
3.2. formation
3.2.1. cation
3.2.1.1. metal
3.2.2. anion
