corrosion
by nick stuart
1. electrochemical corosion: operates in cells like in a car door
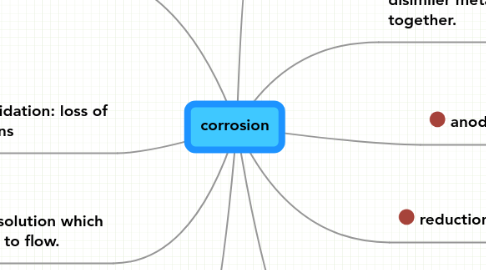
2. electrolytes: solution which allows electrons to flow.
3. oxididation: loss of electrons
4. cathode: reduces
5. galvanic attack: when disimiler metals are placed together.
6. direct chemical attack: creates a uniform film over the surface
7. reduction: gain of electrons
8. corrosion is the chemical detirieration of a matirial.
9. anode : oxidates


